MS, also known as disseminated sclerosis or encephalomyelitis disseminata, is a disorder in which the myelin sheath around the axons of our brains and spinal cord is damaged, leading to demyelination and scarring. In addition, a comprehensive spectrum of indications and symptoms is commonly referred to as Multiple sclerosis. Autologous hematopoietic stem cell transplantation can be life-changing.
What Is MS?
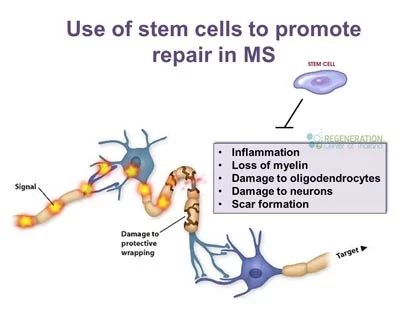
Over time, MS changes the ability of nerve cells, immune cells, and the spinal cord to communicate with each other. Nerve cells generally communicate by sending electrical signals down long fibers known as axons. Axons are wrapped in an insulating 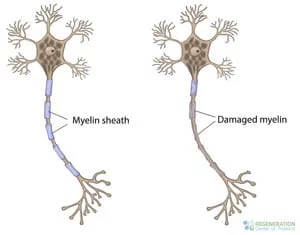 bio-material called myelin. In MS, our body’s immune system attacks and damages the myelin sheath. When our myelin is lost, axons can no longer efficiently conduct signals or communicate as usual, a significant issue for people with progressive MS.[1]
bio-material called myelin. In MS, our body’s immune system attacks and damages the myelin sheath. When our myelin is lost, axons can no longer efficiently conduct signals or communicate as usual, a significant issue for people with progressive MS.[1]
Sclerosis refers to scars, plaques, or lesions in the white matter of our brain and spinal cord, primarily composed of myelin. Although we know much about the mechanisms and symptoms of this degenerative disease, the causes of autoimmune multiple sclerosis MS vs myasthenia gravis remain relatively unknown. Several neurological and immune system symptoms can appear with MS disorder, and frequently, the symptoms progress to display severe physical and cognitive impairment. New symptoms for people with MS can occur rapidly or can occur slowly after related illnesses such as transverse myelitis. The slow appearance of symptoms is known as Multiple progressive sclerosis. The symptoms of MS can sometimes go away entirely on their own. Still, they can also lead to irreversible neurological degeneration seen in movement disorders, spinal cord injuries, Parkinson’s disease, Wernicke’s Aphasia, and particularly in Primary Progressive Multiple Sclerosis.[2] Neurological testing can help identify the disorder whose primary features are standard in various forms of MS: myelinolysis in patients’ brains and spinal cord. The demyelinating disease primarily affects the “CNS” or central nervous system. A well-known type of demyelinating disease is known as Multiple sclerosis or MS.[3]
Categorization of MS & Balos Disease:

- MS or Multiple Sclerosis
- Acute Multiple Sclerosis – Progressive MS
- Persistent cerebral cord sclerosis
- Balo concentric diffuse cerebral sclerosis
- Diffuse cerebral sclerosis – Schilder’s disease
- Inflammation of Optic nerve myelin – Devic’s disease
- Acute disseminated encephalomyelitis
- Necrotizing hemorrhagic encephalitis – Sub-acute and Acute associated with forms of MS
4 Variations of Multiple Sclerosis
- RRMS or Relapsing-Remitting Multiple Sclerosis (relapsing-remitting ms): The most common type of MS we treat. RRMS symptoms include repeat attacks with progressive loss of nerve functions after each attack, followed by a period of remission where the average function is partially or restored without flare-ups or Exacerbations.
- SPMS or Secondary Progressive Multiple Sclerosis Stage: Secondary progressive MS or SPMS usually follows RRMS after continued attacks. This stage results in progressive neurological damage that is associated with the death of motor neurons and nerve demyelination, which is often observed in progressive forms of MS.
- PPMS or Primary Progressive Multiple Sclerosis MS: This stage represents a steady decline with no intermittent remissions, exacerbations, or Flare-Ups
- PRMS or Progressive Relapsing Multiple Sclerosis Stage: PRMS is the rarest type of MS that affects people and is the most difficult to treat. Most patients will not qualify for stem cell transplants, and the patients that are usually recommended an aggressive cell protocol as part of a combination therapy that includes Immunosuppressive techniques to have a positive effect and slow down the rapidly progressing condition caused by MS.
MS is amongst the most severe and complicated nervous system disorders to treat that we have today in the field of regenerative medicine. MS Disorder frequently invades specific regions of the human nervous system, causing intense exhaustion along with other relapsing MS symptoms and clear physical signs that can be readily identifiable using radiology scans such as MRI scans.
Clinical indications of MS include:
- Nystagmus
- Motor weakness (sometimes misdiagnosed as fibromyalgia )

- Visual impairments
- Limb paresis and strokes can be devastating, but stem cells directly applied may assist in recovery. Or paralysis (sometimes misdiagnosed as systemic lupus erythematosus)
- Avascular necrosis
- Orthopedic and hematopoietic stem cell transplantation procedures often aim to alleviate severe conditions. Hematopoietic stem cell transplantation procedures often aim to alleviate severe conditions. Shoulder Injuries
- Diplopia and damage to nerve cells
- Spondyloarthropathy
- Peripheral Neuropathic Pain
- Ataxia
- Motor Neuron Disease MND
- Bladder dysfunction and urinary incontinence or IBD
Lab evaluation for people with MS or relapsing MS usually requires detailed cytology of the patient’s cerebrospinal fluid, along with detailed protein analysis and Radiographic MRI scan examinations, recommended by the national MS society,, recommended by the national MS society, for areas around the encephalocele. Genetic tests and Radiographic exams look for multiple or isolated lesions in the brain. Stem cell research shows that over 65% of the patients treated for MS using neural progenitor cells, undifferentiated cells, interferon Beta, and HSCT (Hematopoietic stem cells) show improvement shortly after treatment. In contrast, some patients show continuing improvement over a few years.
Mesenchymal Stem Cells for Multiple Sclerosis
Stem cells are (undifferentiated/undefined) cells with the ability of perpetual self-renewal and unique properties to produce specialized cells through cell division. Stem cells are usually categorized into embryonic or adult stem cells. Embryonic stem cell lines (ESC) are pluripotent and can give rise to all cell types in the body and differentiate into different tissue types. Embryonic cells are acquired from the inner cell mass of a blastocyst but can lead to serious adverse events in people with progressive forms of MS. Due to strict safety standards, The Regeneration Center does not offer human embryonic stem cells or iPSC cells.
What Are Stem Cells
Adult stem cells are used in the treatment of amyotrophic lateral sclerosis. Progressive MS can begin the healing process by differentiating into many other cell types and is considered crucial in stem cell treatments such as autologous hematopoietic stem cell transplantation. Multipotent. The primary purpose of adult stem cells & immune stem cells is to treat and repair the body using specialized cells needed to reduce inflammation. This need usually arises due to wear and tear, acute injury, disease, and infections. Adult stem cells, including immune cells, can be found in many organs and damaged tissues in the body and are further divided into different categories depending on where they are found. Adult stem cells are either:
Mesenchymal stromal cells and growth factors needed to improve essential functions can be found in several areas, including those undergoing hematopoietic stem cell transplantation and mesenchymal stem cell therapy. 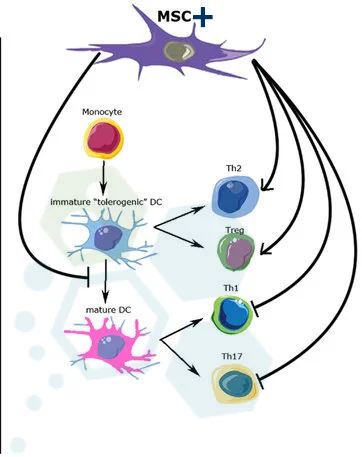
- Bone marrow (BM)
- Peripheral blood
- Umbilical cord blood
- Umbilical cord tissue stem cells
- Dental Pulp – Dental-derived MSCs (DMSCs)
- Placental Tissue / Wharton’s Jelly
Mesenchymal stem cells have unique properties and can be cultured & differentiated (UC-MSC+) into many types of new cells, including:
- Cartilage Cells
- Fat cells
- Skin cells
- Bone Cells
- Ligament
- Tendon Cells
- Muscles cells
- Nerve cells
More recently, reprogrammed pluripotent stem cells known as Induced pluripotent stem cells (iPSCs) offer potential therapies for people with secondary progressive MS. Induced pluripotent stem cells have similar features to embryonic stem cells; however, they carry the risk of tumor formation. This cancer risk can be reduced via transdifferentiation; however, the results have not been consistent or safe. Progenitor stem cells such as induced oligodendrocyte progenitor cells (iOPC), induced neural stem cells (iNSC), and oligodendrocytes (iOL) can also be derived from somatic adult cells, which can be used in stem cell treatment, type via the process of transdifferentiation utilized in stem cell treatment
Effective treatment for MS – HSCT
Hematopoietic stem cells have pluripotential and self-renewal ability but are only found in less than 0.01% of all nucleated cells in the Bone Marrow. Hematopoietic stem cells are capable of forming all hematopoietic cell lines, including:
- Megakaryocytes
- Platelets play a crucial role in recovery after autologous hematopoietic stem cell transplantation.
- Erythrocytes
- Adaptive immune system cells
- Innate immune system cells
Isolated Hematopoietic cells (HSC+) can lead to self-renewal to regenerate damaged nerves using hematopoietic cell types when the new cells are transplanted into patients. HSC cell transplantation (HSCT) has been used for over 50 years as a therapeutic tool to fight cancer, high-dose immunosuppressive therapy to avoid the risk of MS relapses, and Rebuild the immune system after chemotherapy using a type of stem cell treatment known as autologous hematopoietic stem cell transplantation.
Disease-modifying therapies to treat multiple sclerosis
Clinical studies and peer-reviewed studies have shown that hematopoietic stem cells & autologous hematopoietic stem cells are also effective in repairing nerve-insulating myelin caused by various autoimmune conditions, including autoimmune encephalomyelitis (experimental autoimmune encephalomyelitis) have become a viable alternative therapy (with an alternative to immunosuppressive and immunomodulatory medications needed in autoimmune diseases such as MS. Results are achieved by several ways including rebooting the immune system and elimination of autoantigen-reactive T and B lymphocytes along with increasing the populations of t regulatory cells,
How is Effectiveness Measured?
The treatment effectiveness for MS therapies is measured in several ways, including:
- Relapse-free survival rate
- Progression-free survival rate
- MRI event-free survival
- Disease-free survival
- Sustained reduction in disability EDSS score
New treatment for MS – Mechanisms of Action
Double-blind stem cell research has shown that UC-MSC+ mesenchymal cells act as catalysts for improving Inflammatory diseases, including active relapsing MS, which can be challenging to manage. Via several mechanisms, including:
- Immunomodulation
- Immunosuppression
- Neurotrophic repair functions from neural cells for neurological disorders
Long-term benefits of UC-MSC+ stem cells for Neurological conditions such as relapsing-remitting MS can significantly impact daily life. This can include inhibited adaptive immune responses, stopping disease progression, creating new white blood cells, repairing nerve insulating myelin, inhibiting innate immune responses, and proliferation of regulatory t cells, white blood cells, B cells & stem cells found in various tissues. NK cells, a type of cell found in the immune system,
Is Multiple Sclerosis Genetic Testing Available?
MS is generally not considered a hereditary disease. Still, there are over 100 genetic markers that have been identified as increasing the risk of getting MS. Clinical trials, mRNA vaccines, which hold potential, are being reviewed by the MS Society., and gene therapies are being developed to target genetic defects using several gene-editing methods including zinc finger nucleases, transcription activator-like effector nucleases (TALENs), and the clustered regularly interspaced short palindromic repeats (CRISPR)/Cas9 systems. Gene testing is still very early; however, the early patients showed improvement. The goal of these new promising alternative treatments for MS would be to use the combination of stem cell therapy after completing genome editing or mRNA vaccines, which are currently in clinical trials.
Treatment for Progressive MS w/ Stem Cells in 2025
The Regeneration Center has developed a multi-stage program to treat MS. Nerve Cells, nerve fibers, exosomes, Brain stem cells, and neurons can properly regenerate damaged areas of nerve myelin and axons. In contrast, mesenchymal stem cells help to reverse any damage caused by autoimmune disease. This alone can slow down, stop, or even reverse the progressive nature of MS. At the same time, enriched neural cells, glial cells, astrocytes, and neural progenitor cells are used to target the existing brain lesions.
TREATMENT RISKS & PRECAUTIONS
Please note that not all patients are suitable candidates for treating Multiple Sclerosis (MS), Primary Lateral Sclerosis (PLS), or undergoing Hematopoietic Stem Cell Transplant (HSCT). Patients with advanced neurological damage, degenerative Neurological conditions, severe disability, recent heart attack, or other significant health complications might not be good candidates for treatment.1 Yr After Stem Cell Therapy for Progressive Multiple Sclerosis
MS Stem Cell Therapy Overview
The total number and types of stem cells used to treat MS, as evaluated in a cell therapy trial, show promise for MS treatment. It depends on the patient’s medical needs.
Type of Injections: For patients with progressive MS and late-stage MS, treatment of multiple sclerosis may require more aggressive combination therapy using different types of stem cells, including hematopoietic or umbilical cord stem cell transplants (UC-MSC). To ensure long-term efficacy, the cell infusions are done over multiple dates and do not require invasive surgery. The cells are commonly delivered Intravenously through stem cell nebulizers, Intrathecal Injections, or fluoroscopy guidance delivery (in a hospital setting only) of stem cells to bypass the blood-brain barrier.
Physical Rehabilitation Post-Therapy: Physical Rehabilitation therapy for people with relapsing MS is optional, depending on the patients’ travel/time constraints. Complete physical rehabilitation post-therapy can be provided upon request for 2-5 hours per day and up to 5 days per week. Medical visas and extended stay accommodations for the patients and families can also be included upon request. Learn about a multiple sclerosis-friendly diet.
Cost of Treatment for MS with Stem Cells – 2025
Due to the varying degrees of existing medical conditions (eds scores) and the stage of degeneration, our medical team will need to review a potential candidate’s current medical information before a treatment protocol can be established without increased risk. Upon acceptance, we can provide a more accurate assessment of the patient’s condition and what we expect to achieve post-therapy. The final plan will include a daily treatment outline with the total nights required and the complete and fixed medical-related expenses (excluding accommodations or flights). Stem Cell Therapy for MS will require roughly 14 days in Bangkok. To begin evaluating our multi-stage MS Treatment protocol, please prepare your recent medical records, such as Brain MRIs, CT Scans, or PET Scans, and contact us today.
To learn more about treating MS with isolated UC-MSC+ stem cells, please get in touch with us today.
Published Clinical Citations
[1] ^ Pipatpajong, Hemmarin, and Kammant Phanthumchinda. 2011. Neurofibromatosis type I associated multiple sclerosis can manifest in people with secondary progressive MS. Journal of the Medical Association of Thailand, Chotmaihet thangphaet, no. 4. https://www.ncbi.nlm.nih.gov/pubmed/21591539
[2] ^ Laosanguanek, Naressak, Thaddao Wiroteurairuang, Sasitorn Siritho, and Naraporn Prayoonwiwat. 2011. Reliability of the Thai version of SF-36 questionnaire for evaluating quality of life in multiple sclerosis patients in a multiple sclerosis clinic at Siriraj Hospital. Journal of the Medical Association of Thailand = Chotmaihet thangphaet. https://www.ncbi.nlm.nih.gov/pubmed/21721432
[3] ^ Hou, Zong-liu, Ying Liu, Xi-Hong Mao, Chuan-yu Wei, Ming-yao Meng, Yun-hong Liu, Zara Zhuyun Yang, et al. 2013. Transplantation of umbilical cord and bone marrow-derived mesenchymal stromal stem cells in a patient with relapsing-remitting multiple sclerosis ms, progressive ms with neural cells adhesion & migration, no. 5 (October 30). doi:10.4161/cam.26941. https://www.ncbi.nlm.nih.gov/pubmed/24192520