We recently experienced the powers of a ground-breaking vaccine against COVID-19 thanks to mRNA technology, but it has even more potential to democratize access to bleeding-edge medical advancements. Messenger RNA or “mRNA” is a single-stranded RNA involved in protein synthesis. Although the blueprint for proteins in living cells was discovered six decades ago, its pharmaceutical potential was long overlooked. mRNA degradation prevention didn’t look promising; it was too unstable, not very effective, and inflammatory [1].
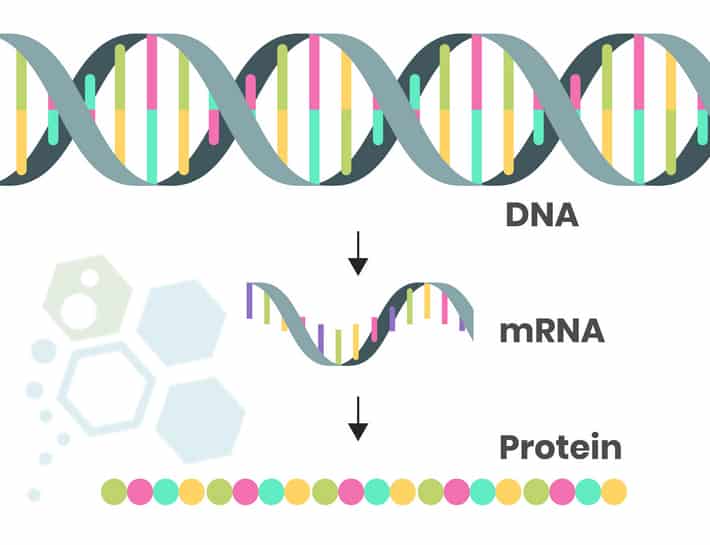
What does mRNA do?
Each mRNA messenger carries instructions to make a specific protein. These instructions act like a blueprint. mRNA then delivers these instructions for cells to put the protein together in a process known as transcription. mRNA messengers carry protein building information from DNA in the cell’s nucleus all the way to the cell’s watery interior (cytoplasm), where our protein-producing machines read the mRNA sequences and translates each three-base mRNA codon (nucleotide triplets) into its corresponding amino acid inside a growing protein chain.
The future of mRNA
History of mRNA
Researchers have been experimenting with mRNA-based vaccines and treatments for several decades already. The first treatments were performed on rodents back in the early 1990s, for several diseases including diabetes and cancer. A first in medical history, the successful creation of the first mRNA vaccines against Covid-19 in 2020 was hailed as a triumph. The rapidly iterative advancements we mostly fueled by the independent contributions of scientists and researchers from all over the world. Because of its adaptability, capacity to activate the immune system, and its safety profile. After the activated mRNA sequence perform their biological function, the molecule is said to completely degrade, leaving no trace in the body [3].
The ability to deliver mRNA molecules to the proper immune cells in the body, as well as to increase its stability and effectiveness, were all things the medical world learned how to do. This 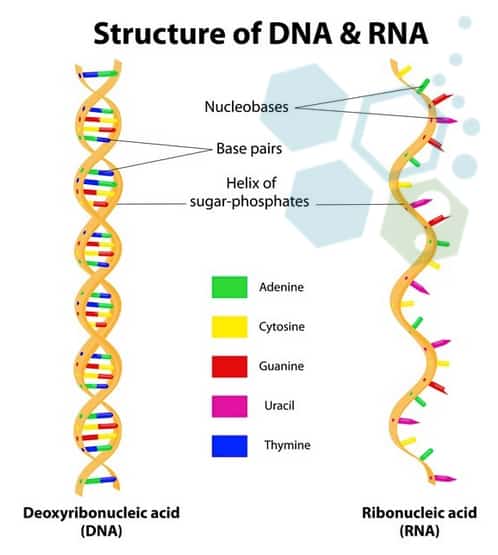 advancement made it possible to develop potent mRNA to amino acid vaccines that, when given to humans in small doses, trigger strong immune responses. Furthermore, researchers are able to develop quick, scalable procedures to produce fresh vaccine candidates for clinical use in a matter of a few weeks.
advancement made it possible to develop potent mRNA to amino acid vaccines that, when given to humans in small doses, trigger strong immune responses. Furthermore, researchers are able to develop quick, scalable procedures to produce fresh vaccine candidates for clinical use in a matter of a few weeks.
mRNA Therapy
Beyond the coronavirus, mRNA lipids have many other applications. Malaria, Dengue fever and tuberculosis are some of the oldest and deadliest pathogens in the world, and doctors are learning to use this technology to combat them more effectively. Each year, there are over 9 million cases of tuberculosis worldwide.[2] The medical need is even greater for malaria because some reports estimate over 230 million malaria cases each year, with the majority of deaths occurring in children under the age of five.
mRNA technology: Advantages and limitations
Thanks to the new research we now have the capacity to identify ideal vaccine targets is enhanced by the convergence of medical innovations, from next-generation sequencing to modifying stem cells for use in therapies we have made giant made strides in better understanding the pathogens that cause bacterial attacks that lead to tuberculosis, den and malaria, with insights into how to combat them. Modeling of proteins’ three-dimensional structures is now possible thanks to the ongoing revolution in computational protein structure prediction models. This enables researchers to identify the parts of these proteins that make the best targets for the creation of vaccines[4].
Do mRNA Vaccines Contain Human Cells?
The Pfizer and Moderna pure mRNA synthesis do not not use any stem cells. The vaccines from AstraZeneca and Johnson & Johnson vaccines use adenovirus as a vehicle to deliver spike protein genes to the cells and these viruses need to grow in those cells. After culturing the adenovirus it is harvested from the cells and then purified to contain only the virus vehicle and does not contain any human cells and do not increase risk of strokes. The initial cell line for AstraZeneca did come from human kidney cells. However, they were obtained over 50 years ago and the current iteration does not contain any human cells.
mRNA-Based Genetic Reprogramming
mRNA technology allows quickly testing hundreds of potential vaccine targets. Furthermore, we can combine various mRNAs, each of which encodes a distinct pathogen antigen, into a single vaccine. For the first time in history, its possible test for genetic abnormalities and train the human immune system to combat numerous weak points in a pathogen by using an mRNA-based vaccine. The first mRNA vaccine candidates against malaria and tuberculosis that combine well-known and novel targets are expected to start clinical trials later this year. If successful, the effort might alter how we prevent these diseases and eventually help to eradicate them.
mRNA splicing treatments are also being used in targeted cancer treatments for:
- Kidney Cancer
- Lung Cancer
- Liver Cancer
- Pleural mesothelioma
- Pancreatic Cancer
- Prostate Cancer
- Colorectal Cancer
- Multiple Myeloma
The benefits of using mRNA cancer therapies are that they can be tailored exactly to individual patient needs by triggering a desired response by the patients immune system to attack the tumor cells in the body without the harmful side effects of traditional chemotherapy.
Other mRNA vaccine based treatments that are currently being developed include:
- HIV
- RSV (respiratory syncytial virus)
- Zika Virus
- Epstein-Barr virus
- Cystic Fibrosis
- Shingles
- Parkinson’s Therapy
- Flu
Thanks to the recent advancement, mRNA technology will be accessible to everyone worldwide and have a direct impact on people’s daily lives.
Published Clinical Citations
[1] ^Qin S, Tang X, Chen Y, Chen K, Fan N, Xiao W, Zheng Q, Li G, Teng Y, Wu M, Song X. mRNA-based therapeutics: powerful and versatile tools to combat diseases. Signal Transduct Target Ther. 2022 May 21;7(1):166. doi: 10.1038/s41392-022-01007-w. PMID: 35597779; PMCID: PMC9123296.
[2] ^Saita S, Maeakhian S, Silawan T. Temporal Variations and Spatial Clusters of Dengue in Thailand: Longitudinal Study before and during the Coronavirus Disease (COVID-19) Pandemic. Trop Med Infect Dis. 2022 Aug 8;7(8):171. doi: 10.3390/tropicalmed7080171. PMID: 36006263; PMCID: PMC9414305.
[3] ^Coppin L, Leclerc J, Vincent A, Porchet N, Pigny P. Messenger RNA Life-Cycle in Cancer Cells: Emerging Role of Conventional and Non-Conventional RNA-Binding Proteins? Int J Mol Sci. 2018 Feb 25;19(3):650. doi: 10.3390/ijms19030650. PMID: 29495341; PMCID: PMC5877511.
[4] ^Fan XY, Lowrie DB. Where are the RNA vaccines for TB? Emerg Microbes Infect. 2021 Dec;10(1):1217-1218. doi: 10.1080/22221751.2021.1935328. PMID: 34036874; PMCID: PMC8216257.