Congestive Heart Failure, or “CHF,” is a state in which the heart cannot properly function as a pump. As a result of the cardiac malfunction, the oxygen pumped into the body is insufficient. Simultaneous illnesses generally cause congestive heart failure.
What is CHF?
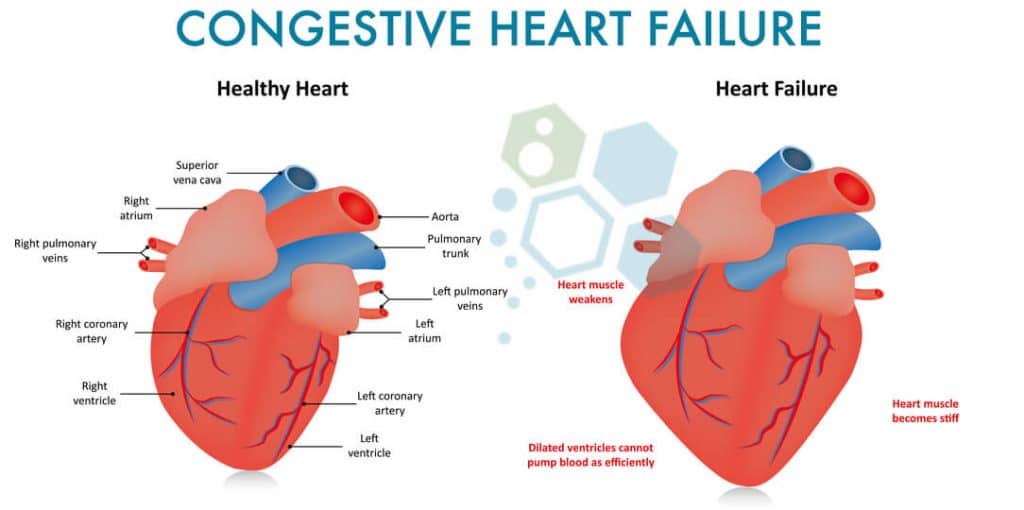
 CHF, or Chronic heart failure, is a progressive condition affecting heart muscles’ power and efficiency. Often referred to as heart failure, this type of cardiovascular disease is diagnosed after fluid buildup around the heart, causing it to function inefficiently. There are two halves and four chambers in the human heart. The heart’s upper half has (2) atrium, while the lower half has (2) ventricles that pump blood throughout the body’s tissues and organs. The atria then receive the blood after a loop to circulate it back.
CHF, or Chronic heart failure, is a progressive condition affecting heart muscles’ power and efficiency. Often referred to as heart failure, this type of cardiovascular disease is diagnosed after fluid buildup around the heart, causing it to function inefficiently. There are two halves and four chambers in the human heart. The heart’s upper half has (2) atrium, while the lower half has (2) ventricles that pump blood throughout the body’s tissues and organs. The atria then receive the blood after a loop to circulate it back.
If the ventricles become damaged (diastolic heart failure) or diseased, blood and other fluids can leak from capillary blood vessels to areas including the liver, lungs, stomach, and legs, creating a life-threatening situation that often requires immediate medical attention. Other symptoms of damage to the heart ventricles include frequent shortness of breath, swelling (edema), and weakness.
Right-Sided vs. Left-Sided Heart Failure
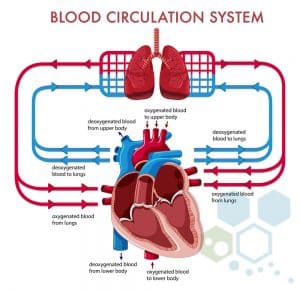
The left side of the heart pumps blood, while the right side pumps blood to the lungs. When blood enters
The right atrium (via vena cava) flows to the right ventricle, pumping it into the lungs via the pulmonary artery. The pulmonary artery carries the deoxygenated blood to the lungs, where oxygen infuses with red blood cells before returning to the left atrium via the pulmonary veins. From the pulmonary veins, blood flows to the heart’s left ventricle before being pumped back to tissues and organs. The red blood cells carry oxygen to our organs while the waste product of carbon dioxide is picked up and removed via the lungs (breath out.) Blood can then return to the right atrium to do the processes over and over again. The pulmonary veins are used to carry oxygenated blood, while the pulmonary arteries are responsible for taking deoxygenated blood. This relationship between veins and arteries is opposite in the rest of the body.
Heart disease is described as an Illness that weakens the cardiovascular system or heart muscle, illnesses that trigger the heart muscles to become stiff, or disorders that create an increase in oxygen demands. These consequently increase the supply of fresh oxygen when the heart is incapable of producing oxygen-rich blood at the needed level.
Congestive heart failure and ischemic heart disease can impact numerous organs in the body. For instance, the injured areas of the heart directly affected by the sickness cannot produce enough blood for the kidneys, which then affects their ability to excrete water and salt (sodium). The distressed kidney function may cause the person to retain more fluids than needed. Patients with higher cardiometabolic risks or a history of muscular atrophy may also develop pulmonary edema (PE).
PE occurs when the fluid in the lungs diminishes a person’s ability to exercise regularly. Fluid might likewise accumulate inside the liver, directly affecting its function by impairing the liver’s ability to create essential proteins and helping clear the system of harmful elements or toxins. The intestines might even be less effective in absorbing the vitamins, nutrients, and medicines a human needs. The fluids can also accumulate quickly, resulting in peripheral neuropathy or edema (severe swelling) of the ankles and feet.
Stages of Congestive Heart Failure
There are four main stages of Systolic heart failure: 1, 2, 3, and 4 or ( A, B, C, D depending on which country)
- Stage 1 is the lowest severity, and they often are unaware of any problems. Stage 1 or pre-heart failure patients are considered at high risk of developing heart failure. Examples of Stage 1 diagnosis include patients with diabetes, History of alcohol abuse, high blood pressure, or coronary artery disease.
- Stage 2 Systolic heart failure is considered pre-heart failure and is usually given to patients with an ejection fraction (EF) percentage of 40 or less, along with systolic left ventricular dysfunction.
- Stage 3 Heart Failure patients are previously or currently diagnosed with diastolic heart failure due to the underlying structural damage to the heart and cardiac function. Patients diagnosed with stage 3 (C) of heart disease are usually prescribed Beta-blockers, Hydralazine/nitrate combinations, vasodilator medicines (Aldosterone antagonist), heart stem cell repair, diuretics, and may need implantable cardiac defibrillator (ICD) or biventricular pacemaker.
- Stage 4 Heart Failure is considered the final (end) stage of heart failure. Stage 4 (D) patients typically had a failed treatment in the earlier stages. They required more advanced options, including Open Heart surgery, cardiac stem cell treatments, Ventricular assist devices, continuous inotropic medications, or a heart transplant.
Causes of Diastolic Heart Failure
Cardiomyopathies and damage to the heart can occur due to many reasons, including lifestyles, environment, and genetics. Autoimmune diseases such as psoriasis, rheumatoid arthritis, and lupus can double the risk of heart failure. Some other common risk factors, including
- Diabetes and Diabetic Nephropathy
- Hypertension
- Parkinson’s
- Kidney Failure
- Lung disease, IPF & COPD
- Viral Infections
- Abuse of drugs & alcohol
- Connective tissue disorders such as systemic lupus erythematosus
- Obesity
- Exposure to toxins such as cobalt & lead
- Reaction to prescription medications

Is CHF Hereditary?
Most cases of heart failure are not hereditary, but some uncommon types of heart disorders can be passed down through generations. Examples of genetic variants include:
- Aortopathy & connective tissue disorders
- Pulmonary arterial hypertension
- Arrhythmia
- Familial Cardiomyopathy (hypertrophic)
- Congenital heart failure
- Hereditary hemorrhagic telangiectasia
- Familial hypercholesterolemia
The Thailand Regeneration Center offers a comprehensive list of DNA tests for heritable heart and vascular conditions, including the following panels.
DOLK,TAZ, TCAP, TMEM43, TNNC1, TNNI3, JUP, LAMP2, LMNA, MYBPC3, DSC2, DSG2, DSP, EMD, MYH7, MYL2, MYL3, PKP2, PLN, PRKAG2, RAF1,EYA4,ABCC9, ACTC1, ACTN2,FHL1, FKRP, FKTN, FLNC, GAA, GLA, HCN4, AGL, BAG3,TNNT2, TPM1, TTN, TTR, VCL,RBM20, RYR2, SCN5A, SGCD, SLC22A5, CACNA1C, CAV3, CRYAB, CSRP3, DES, ANKRD1, CTNNA3, LDB3, PDLIM3 and DMD.
Several randomized clinical trials and stem cell research are underway to treat genetic heart defects using gene therapy; however, none are approved for clinical use yet. Studies have proven that making changes to your diet and getting regular exercise can help reduce the risk of developing end-stage heart failure.
Stem Cell Treatment for Heart Failure
An Ejection fraction (EF) of 20% would be considered a dangerous level and, therefore, indicates a highly advanced stage of heart failure. Healthy people without cardiovascular diseases usually have between 52% and 68% ejection fraction.
The primary issue is that the chamber responsible for pumping one’s heart (The left ventricle) enlarges and isn’t beating effectively, causing the EF percentage level to dangerously low. Ideally, EF percentages ought to be between 52% and 74%. This number also represents the amount of blood pumped from the heart on every beat. The walls of the left ventricle are hypokinetic. The hypokinetic state indicates they are not contracting correctly. This is also why the EF% is low, and the patients have Diastolic Dysfunction of the heart.
Regeneration Center Patient 1 Year After Stem Cells for Heart
Endogenous Cell Therapy for CHF
MSC Stem cells are unspecialized cells with two essential characteristics that distinguish them from other cells inside our bodies. Stem Cells can replenish their numbers naturally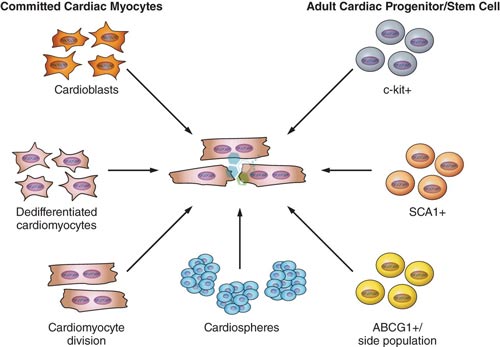 and indefinitely via cell division and the paracrine cell communication mechanism. When circulating stem cells receive chemical indicators of trauma, dysfunctions, or damage, they can migrate directly to the injured/dysfunctional area and transform into specialized cells needed to perform particular functions. Functions or tissues or cells like nerve cells, circulating progenitor cells, mononuclear cells, mesenchymal stem cells, and hematopoietic progenitor cells. Other cardiology therapies include:
and indefinitely via cell division and the paracrine cell communication mechanism. When circulating stem cells receive chemical indicators of trauma, dysfunctions, or damage, they can migrate directly to the injured/dysfunctional area and transform into specialized cells needed to perform particular functions. Functions or tissues or cells like nerve cells, circulating progenitor cells, mononuclear cells, mesenchymal stem cells, and hematopoietic progenitor cells. Other cardiology therapies include:
- Coronary artery disease (CAD)
- Angina
- Biventricular failures
- Disorders of the Heart Valves
- Cardiomyopathy
- Ischemia & Chronic ischaemic heart disease (IHD)
- Terminal Heart failure
- Myocardial infarction – Heart Attacks
Heart Regeneration Therapy Guidelines for 2025
Using stem cells for heart & cardiac regeneration therapies using cord tissue cells is a breakthrough treatment that requires enriched mesenchymal stem cells (MSC+) to be introduced into damaged heart tissue. This cellular proliferation helps enhance the performance/condition of patients suffering from congestive severe heart failure or brain strokes—stem cells for CHF work primarily through creating new blood vessels. Angiogenesis also helps improve tissue perfusion. The freshly expanded cardiac cells are carefully “transplanted” back into the patient through targeted infusions or into the damaged heart muscle or even by infusing them into the coronary arteries. Through the process of “Homing” [2], Stem cells migrate into the heart muscles for months after the initial treatment to assist in the regeneration process through the transformation into healthy functioning heart cells and reverse coronary heart failure.
Stem cell treatment for low ejection fraction
- Sustained improvement in heart pumping power and its ability to regulate the proper supply of blood to the body [3]
- Improved regulation of O2 (Oxygen) Intake and Use [4]
- Increase in myocardial performance, neurohumoral activations, and functional heart capacity
- More significant Reduction in Angina [1]
- Reduction in the infarct size with a decrease in heart muscle scar and reversal of cardiogenic shock
- Long-term follow-up (≥12 months) shows noticeable improvement in the patient’s overall well-being and physical endurance.
- Improvement of ventricular ejection fraction resulting in a reduced need for rehospitalization [5]
CLINICAL TREATMENT PRECAUTIONS & RISKS
Please note stem cell treatment for CHF and cardiac stem cell therapies may not be appropriate for all conditions. Cell therapy, heart tissue, and cell engineering are limited based on the extent of the heart muscle and tissue damage and the patient’s current and accurate medical condition. Patients who are considered “high risk”, have severe inflammatory osteoarthritis or requiring sustained oxygen assistance preventing international travel may not qualify. MSC+ Cardiac therapy is appropriate for patients looking to improve ventricular performance, quality of life, and overall survival rates for patients with heart failure.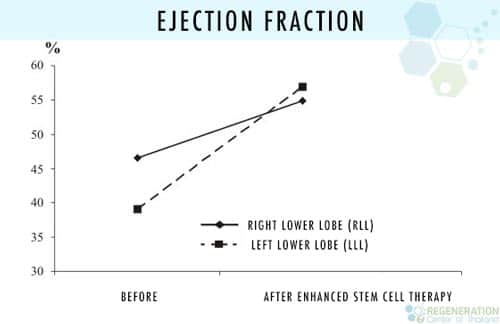
Chronic Heart Failure Treatment Options
Adjuvant therapy using Cardiopoietic MSC+ Stem cells (per treated stage) Severe cases with multiple commodities may require a multiple-stage delivery using transvascular injections with direct injection Into the ventricular walls to ensure optimal results. Rehabilitation after treatment is optional based on the patient’s time constraints. Complete rehabilitation and nursing care can be provided 2-4 hours per day and up to 4 days per week
. Medical visas and accommodations for the patient and family/friends at an extended-stay hotel can be included upon request. Depending on stage and severity, the estimated treatment length is two weeks.
CHF Therapy Requirements & Safety Advice
Our clinical cell therapy for heart failure is focused on providing measurable results that require proper extraction and expansion of MSCs and cardio-sphere-derived cells before infusions. The multi-stage cardiac regeneration protocol is a 2-week procedure with the option of cardio-physical rehabilitation in Bangkok, Phuket, or Chiang Mai for 5-15 days after being treated. Due to the varying degrees of heart disease, our medical team will first need to understand the patient’s current medical needs better to determine whether cell therapy will be beneficial. A detailed treatment plan will be provided upon the medical evaluation. It will include a day-by-day itinerary outlining the cardiac cell regeneration protocol and other details, such as the total number of nights required and the total and fixed medical-related costs. To begin your evaluation for the regeneration center MSC+ heart therapy, please prepare evidence or recent medical records such as an Echocardiogram, EKG, Stress test, Tilt Table, Heart CT Scan, or SPECT exam (Single-photon emission computed tomography) and contact us today.
Published Clinical Citations
[1] ^ Boonbaichaiyapruck, Sarana, Pavit Pienvichit, Thosapol Limpijarnkij, Pairoj Rerkpattanapipat, Apichai Pongpatananurak, Ratchanee Saelee, Artit Ungkanont, and Suradej Hongeng. 2010. Transcoronary infusion of bone marrow derived multipotent stem cells to preserve left ventricular geometry and function after myocardial infarction. Clinical cardiology Thailand, no. 7. doi:10.1002/clc.20545. https://www.ncbi.nlm.nih.gov/pubmed/20552656
[2] ^ Hou, Jingying, Lingyun Wang, Jieyu Jiang, Changqing Zhou, Tianzhu Guo, Shaoxin Zheng, and Tong Wang. Cardiac stem cells and their roles in myocardial infarction. Stem cell reviews and reports, no. 3. doi:10.1007/s12015-012-9421-4. https://www.ncbi.nlm.nih.gov/pubmed/23238707
[3] ^ Nartprayut, Kuneerat, Yaowalak U-Pratya, Pakpoom Kheolamai, Sirikul Manochantr, Methichit Chayosumrit, Surapol Issaragrisil, and Aungkura Supokawej. Cardiomyocyte differentiation of perinatally‑derived mesenchymal stem cells. Molecular medicine reports, no. 5 (March 4). doi:10.3892/mmr.2013.1356. https://www.ncbi.nlm.nih.gov/pubmed/23467912
[4] ^ Pratumvinit, Busadee, Kanit Reesukumal, Kajohnkiart Janebodin, Nicholas Ieronimakis, and Morayma Reyes. 2013. Isolation, characterization, and transplantation of cardiac endothelial cells in Thailand. BioMed research international (October 27). doi:10.1155/2013/359412. https://www.ncbi.nlm.nih.gov/pubmed/24282814
[5] ^ Sheng, Calvin C, Li Zhou, and Jijun Hao. 2012. Current stem cell delivery methods for myocardial repair. BioMed research international (December 27). doi:10.1155/2013/547902. https://www.ncbi.nlm.nih.gov/pubmed/23509740