Throughout modern history, cancer treatments have been dominated primarily by very cytotoxic pharmaceuticals and chemotherapy. These traditional means of treating cancer that is successful in killing cancer cells, but these toxic medications also destroy many of the body’s healthy cells and neurotransmitters, causing severe side effects. The lone goal of scientists has always been to target & only kill cancer cells and reverse systemic autoimmune disease using a functional medical approach to fighting Cancer.
NK Cell Treatment for Cancer
Natural Killer Cell Cancer Immunotherapy
The human body’s first line of defense against cancer is the natural killer Cells or “NK.” Recent advancements in gene therapies,mRNA technology, regenerative medicine, and non-toxic radiology have helped us get much further than ever before. Our current therapeutic goals have focused on strengthening your body’s natural capacity to fight diseases and cancer.[1]
The Human immune system is always finding ways to prevent cancer by utilizing NK Cells or Natural Killer cells. Our immune system can and does destroy precancerous cells, cancerous cells, and viruses while recognizing them and not harming healthy cells. The Regeneration Center has developed an innovative targeted therapy (Autologous NK cellular immunotherapy) & nk cell activation protocol using the patient’s immune system to fight and destroy cancer.
Our unique activated lymphocyte therapy infuses harvested autologous Enriched NK cells from the patient’s immune system to assault and destroy the invading cancer cells directly. NK Cells or ” T killer cells” are a kind of white blood cell known as lymphocytes. These lymphocytes are part of the congenital immune system in humans.[2]
Allogeneic NK cells
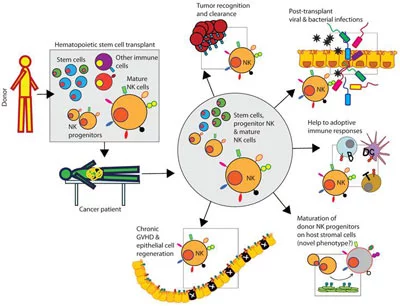
Allogeneic NK cell therapies derived from primary cells blood, cord blood [CB], and cord tissue assist the Regeneration Center with rapid NK cell manufacturing capabilities. Allogeneic (donor-derived NK cells) therapies derived from primary cells such as blood, cord blood [CB], and cord tissue have the advantage of providing a rapid NK cell manufacturing capability. These cell-based therapies involve obtaining primary cells from blood or other tissue sources, isolating and expanding the NK cells in the laboratory, and administering them to patients. Unlike autologous NK therapies, which require the patient’s cells to be harvested and processed, Donor NK Cell (allogeneic) therapies can be manufactured in advance and kept in stock for immediate use by patients with cancer.
NK cell activating
Blood-derived allogeneic NK cell therapies typically involve isolating Blood NK cells from peripheral blood mononuclear cells (PBMCs) obtained from healthy, unrelated donors before the NK cells, feeder cells, car-nk cells, and b cells engineered to express desired receptors. Cord blood-derived therapies, on the other hand, utilize NK cells derived from the blood collected from the umbilical cord and placenta after childbirth. Cord tissue-derived therapies involve isolating NK cells from the tissue of the umbilical cord, and cell-based immunotherapies offer reduced cell cytotoxicity.
NK cell-based immune checkpoint
The advantage of using these primary CAR-T cell sources is that they provide a readily available and renewable supply of NK cells for treatment. Blood and cord blood donations can be obtained from healthy donors, while cord tissue can be collected from the umbilical cords of newborn babies during routine childbirth procedures. This means that many NK cells can be obtained for cell manufacturing without the need for repeated patient-specific cell collection and processing.
Peripheral blood-derived & umbilical cord-derived NK cells
The Enhanced NK cell preparation can take 14-24 days, but the Regeneration Center can begin before patient arrival for allogeneic treatments. Our unique allogeneic NK cell therapies have significant implications for scalability and accessibility. It allows us to produce NK Cells to be stored at our cell bank in Bangkok or prepared (NK cell expansion) for immediate use, reducing the waiting time for patients in need. This is particularly advantageous in critical situations where quick administration of NK cell function is crucial, such as in cancer immunotherapy or treatment of infectious diseases.
Allogeneic NK cell therapies derived from primary cells like blood, cord blood, and cord tissue have a notable advantage of rapid manufacturing capability. This enables the production of many doses that can be readily available for patients, improving accessibility and potentially saving lives with natural killer cells for cancer.
What are the differences between T cells & natural killer cells?
Killer T cells and natural killer cells are immune cells that can eliminate infected or cancerous cells, but they differ in how they recognize and destroy their targets. Killer T cells are T lymphocytes that identify specific antigens on abnormal cells and release toxins to kill them. In contrast, natural killer cells are part of the innate immune system. They can detect and destroy abnormal cells without prior antigen exposure by sensing changes in surface proteins or the absence of a “self” marker. While both cell types contribute to the body’s defense against infected or cancerous cells, they employ distinct target identification and elimination mechanisms.
Treat Cancer Without Chemotherapy, Radiation or Surgery
These Killer cells play a leading part in our body’s ability to reject “foreign mutant cells,” such as tumors or virally infected cells. Human natural killer cells are cytotoxic, immune, or natural killer T cells. Tiny granules within its cytoplasm contain proteins (proteases & perforin) known as granzymes. A large number of cytokines are produced by Natural T Killer cells, including:
- Interleukin (IL-10)
- Tumor necrosis factor α TNFα
- IFNγ
Potential Side Effects of NK cell therapy
NK (natural killer) cell therapy, a promising immunotherapy, involves using the body’s immune system to target and destroy cancer or infected cells. However, like any medical treatment, NK cell therapy can have potential side effects, depending on the patient’s condition, dosage, and individual response. One of the primary risks is cytokine release syndrome (CRS), which occurs when NK cells trigger an overwhelming immune response, leading to symptoms like fever, fatigue, low blood pressure, and, in severe cases, organ damage. This is a known complication in immunotherapies where the immune system becomes over-activated. Another possible side effect is graft-versus-host disease (GVHD), mainly if donor-derived NK cells are used. In this condition, the donor cells attack the patient’s healthy tissues, leading to symptoms such as skin rashes, liver inflammation, or gastrointestinal distress.
Autoimmune reactions may also develop, where NK cells inadvertently target healthy cells or tissues, potentially causing inflammation or damage to organs such as the liver, lungs, or heart. Additionally, patients might experience flu-like symptoms, including chills, headaches, and muscle aches, as their immune system responds to the therapy. There is also a risk of infection, as the immune system’s modulation could lead to temporary immune suppression, making the patient more vulnerable to bacterial, viral, or fungal infections. Patients may have allergic reactions to therapy components, ranging from mild symptoms, like itching or swelling, to more serious anaphylactic reactions.
Long-term side effects are still being studied, as NK cell therapy is relatively new, but careful monitoring and personalized treatment protocols are essential to minimize risks. Despite these potential side effects, the benefits of NK cell therapy, particularly in treating cancers and viral infections, often outweigh the risks for eligible patients.
Enhance NK Cell + Immunotherapy.
Upon expansion of Autologous NK Cells, they are released back into the patient’s bloodstream while near targeted cancer cells for the following:
- Rectal Cancer
- Lung Cancer
- Liver Cancer
- Pancreatic Cancer
- Kidney Cancer & Renal cell carcinoma
- Pleural mesothelioma
- Prostate Cancer
- Squamous cell carcinoma (head and neck squamous cell)
- Ovarian cancer
Future directions for NK cell therapy
Clinical studies that will help us develop future immunotherapies should continue to provide promising strategies to exploit the unique functions of NK cell lines to treat cancer, infections, and other pathological conditions. Below are some future directions and areas of exploration in the realm of Human NK cell therapy:
Improved Cell Sources for Engineered Car-Nk Cells
Off-the-shelf Products: Unlike T-cell therapies like CAR-T, which are patient-specific, NK cells could be used as “off-the-shelf” therapies.
iPSC-derived NK Cells: Induced pluripotent stem cell (iPSC) derived NK cells offer a standardized, scalable cell source.
Advanced Engineering – Activate nk cells:
- Chimeric Antigen Receptor (CAR-NK) Engineering, which involves creating an NK cell line with specific receptors that more precisely target cancer cells, is a growing area of research.
- Multi-specific Targeting: Creating NK cells targeting multiple antigens can increase efficacy and minimize resistance.
- Better Co-stimulatory Molecules: Clinical trials are ongoing to identify which co-stimulatory molecules may help improve the efficacy of engineered NK cells.
TREATMENT RISKS & PRECAUTIONS
Please note that not all patients are suitable candidates for Natural Killer (NK) Cell Treatment, CAR-T cell therapies, or Immunotherapy with NK Cells. Patients with advanced disease progression, severe immune suppression, or other critical health conditions might not be good candidates for the estimated 3-5 week treatment.Enhanced Persistence and In Vivo Expansion of NK Cells:
- Memory-like NK cells: Unlike primary NK cells, some modified NK cells exhibit memory-like properties and may offer prolonged surveillance.
- Combination with IL-15: Using cytokines like IL-15 to improve the in vivo persistence of NK cells for cancer immunotherapy.
- Combinatorial Approaches with NK Cells + Checkpoint Inhibitors: NK cells are combined with dendritic cells and other checkpoint inhibitors, such as PD-1/PD-L1 or CTLA-4 inhibitors.
- Combinatorial Approaches with a Combination with Vaccines: Combining NK tumor cell treatment with cancer vaccines will better prime the immune system.
- Synergy with Chemotherapy or Radiation: Adoptive NK cells may work well when conventional treatments target the tumor.
Tackling Solid Tumors – NK Cell Biology:
While initial studies have often focused on hematological malignancies, future therapies are likely to explore the role of NK cells in combating solid tumors. NK cells also have a role in fighting viral infections and other diseases, offering a vast potential application.
Regulatory and Ethical Considerations:
- Safety Protocols: As with all cell therapies, understanding and minimizing potential side effects will be crucial.
- Cost-effectiveness: New techniques must be developed to make these therapies financially accessible.
- Data Integration with AI and Machine Learning: Utilizing computational methods to optimize NK cell therapy strategies and predict patient responses.
The field of NK cell therapy is likely to see significant advances in the coming years, thanks to a combination of technological innovation, increased understanding of cancer biology, and improved manufacturing practices. To learn more about DNA screening for hereditary cancers with the Regen Center using NK Cells, please contact us today.
Published Clinical Citations
[1] ^ Knorr, David A, Veronika Bachanova, Michael R Verneris, and Jeffrey S Miller. 2014. Clinical utility of natural killer cells in cancer therapy and transplantation. Seminars in immunology, no. 2 (March 5). doi:10.1016/j.smim.2014.02.002. https://www.ncbi.nlm.nih.gov/pubmed/24618042
[2] ^ Mimura, Kousaku, Takahiro Kamiya, Kensuke Shiraishi, Ley-Fang Kua, Asim Shabbir, Jimmy So, Wei-Peng Yong, et al. 2014. Therapeutic potential of highly cytotoxic natural killer cells for gastric cancer. International journal of cancer, no. 6 (February 28). doi:10.1002/ijc.28780. https://www.ncbi.nlm.nih.gov/pubmed/24615495
[3] ^ Pongpruttipan, Tawatchai, Sanya Sukpanichnant, Thamathorn Assanasen, Pongsak Wannakrairot, Paisarn Boonsakan, Wasana Kanoksil, Kanita Kayasut, et al. 2012. Extranodal NK/T-cell lymphoma, nasal type, includes cases of natural killer cell and αβ, γδ, and αβ/γδ T-cell origin: a comprehensive clinicopathologic and phenotypic study. The American Journal of surgical pathology, no. 4. doi:10.1097/PAS.0b013e31824433d8. https://www.ncbi.nlm.nih.gov/pubmed/22314189
[4] ^ Selathurai, Ahrathy, Virginie Deswaerte, Peter Kanellakis, Peter Tipping, Ban-Hock Toh, Alex Bobik, and Tin Kyaw. 2014. Natural killer (NK) cells augment atherosclerosis by cytotoxic-dependent mechanisms. Cardiovascular research, no. 1 (January 26). doi:10.1093/cvr/cvu016. https://www.ncbi.nlm.nih.gov/pubmed/24469537