Telomeres are essentially the “butts” of single cell chromosomes. They are the tail end of a repetitive DNA structure that is highly related to your typical sequence of DNA that has undergone replication over and over again in a very distinct manner. A telomere will first neutralize a chromosomes’ predisposition/pre-programmed instructions to shorten with copy it makes. In other words, Telomeres are what helps to protect the very instructions of life from rapid deterioration that we call aging.[1] This has let to a tremendous amount of interest in trying to manipulate the gene into change the very coding in our chromosomes to further delay or possible even reverse the process of senescence.
What Are Telomeres?
The first known account of the amazing natural phenomenon was reported by Dr Alexei Olovnikov back in March of 1971. Dr Olovnikov later won a Nobel Prize for his findings. Doctor Olovnikov was the first to suggest that a tiny fraction of DNA sequencing is lost in every “copy” and replicative stage until the point when it reaches a critical level. After this level/age cell division would just stop. This stage is known as apoptosis. In cell division and cell differentiation, specific enzymes help copy our chromosome so DNA does not duplicate to the exact end part of a chromosome and can reach a total length of nearly 15,000 base pairs.
Today, we generally believe that the immortal DNA strands somehow avoid Telomere loss. Most scientists around the world believe that healthy Telomere play a critical role in the stability of our chromosomes. Most scientists also believe that the very act of growing old/aging hinges directly on the act of replicative senescence. [2]
End replication problem is a term to describe when cells divide without telomeres. Telomeres’ basically act like little disposable buffers at the end point of chromosomes. [3] These end points are totally consumed during cell division and thereby help to naturally replenish through this enzyme.

Telomerase are also being tested in gene therapies for treating cancers. Cancer cells divide at nearly 15-20 times the rate of normal cells. It is believed that if telomerase activity could be turned off with gene splicing then telomeres in the cancer cells would basically not divide as fast just like help to do in regulating normal body cells. By controlling how fast the cancer cells divide, we would be able to prevent the rapid spread of cancer cells in the early stages of uncontrollable replication.
Telomere have been known to help Reverse transcriptase “RT” and are also used in theraputic cloning. The benefits of the use of telomere in regenerative medicine especially for anti-aging treatments rest on the fact that the entire life activity of telomeres is essentially controlled by only 2 mechanisms: erosion & addition. If we can slow the erosion process we may be able to manage our aging more aggressively than mother nature can.
Telomeres are the protective caps at the ends of eukaryotic chromosomes. Composed of repetitive DNA sequences and specialized proteins, they serve to protect the integrity of the chromosome by preventing degradation, fusion with other chromosomes, and irregular recombination. Essentially, telomeres act like the plastic tips on the ends of shoelaces, keeping chromosomes from fraying and sticking to each other.
Structure and Composition
The structure of a telomere is primarily made up of a simple repeating sequence of DNA. In humans, the sequence is usually TTAGGG. This sequence is repeated hundreds to thousands of times. Telomeres also contain a variety of proteins that protect the chromosome ends and regulate telomere length.
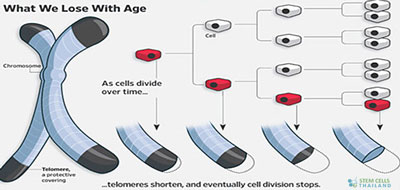
Telomere Role in Cellular Aging
- Replicative Senescence: Every time a cell divides, the telomeres get shorter due to the end-replication problem associated with linear DNA replication. When they reach a critically short length, the cell enters a state called “replicative senescence” where it ceases to divide.
- Cellular Immortality: Some cells, like stem cells and cancer cells, express an enzyme called telomerase that can extend the telomeres, granting these cells a form of “cellular immortality.”
- Implications in Medicine and Cancer Research: Uncontrolled cell division is a hallmark of cancer. Some cancers achieve this by activating telomerase or other mechanisms to maintain telomere length.
- Anti-Aging Treatments: Telomere shortening has been proposed as a biological clock that contributes to aging, although this is still a topic of active research in stem cell treatments for wellness.
- Stem Cell Function: Understanding how telomeres and telomerase work in stem cells can have implications for regenerative medicine.
Current Research and Challenges
- Diagnostic and Prognostic Tool: Telomere length is being studied as a potential biomarker for various diseases including cancer, cardiovascular diseases, and aging-related illnesses.
- Therapeutic Targets: Drugs that can manipulate telomere length or inhibit telomerase activity are being researched for their potential use in cancer therapy.
Can we tap the “Fountain of Youth,” simply with a shot of telomerase?
No. Currently there has not been enough research,clinical tests or practical applications in this area of microbiology. Any company,product or service claiming otherwise is false. Be cautious of any pills, peptides, injections, serums or creams claiming the active involvement of Telomeres. Taking such products is likely dangerous and will lead to more harm than good.
Telomeres play a critical role in cellular function, aging, and disease. Understanding the complexities of telomere biology not only can shed light on fundamental biological processes but also has the potential to revolutionize the fields of medicine and regenerative healthcare.
Published Clinical Citations
[1] ^ Lurdes Planas, Elena Garcia Arias-salgado, Ana Montes Worboys, Pilar Rivera Ortega, Vanesa Vicens-Zygmunt, Roger Llatjos Sanuy, Patricio Luburich Hernaiz, Ignacio Escobar Campuzano, Elisabeth Arellano, Eva Balcells, Julio Cortijo, Ernest Sala, Jordi Dorca, Rosario Perona, Maria Molina-Molina, Biological age instead of chronologic age as prognostic factor in IPF: clinical implications of telomere shortening, QJM: An International Journal of Medicine, Volume 109, Issue suppl_1, September 2016, Page S10, https://doi.org/10.1093/qjmed/hcw124.004
[2] ^ K. Al-Issa, L.B. Tolle, A.S. Purysko, I.A. Hanouneh, Short telomere syndrome and fibrosis, QJM: An International Journal of Medicine, Volume 109, Issue 2, February 2016, Pages 125–126, https://doi.org/10.1093/qjmed/hcv115
[3] ^Ram Naikawadi, Supparerk Disayabutr, Benat Mallavia, Matthew Donne, Gary Green, Jason Rock, Mark Looney, Paul Wolters,Telomere Dysfunction in Alveolar Epithelial Cells Causes Pulmonary Fibrosis: Role for TRF1 , QJM: An International Journal of Medicine, Volume 109, Issue suppl_1, September 2016, Page S67, https://doi.org/10.1093/qjmed/hcw120.032