The exact origin of the term “Daughter Cell” is unknown but it is believed the term comes from the result of asexual binary fission. The cells formation is asexual by definition, if the parent cells are considered to be “mother” cells, then the progeny would have to be of considered the same “gender” or “female daughters”, since the genetic material in the newly formed cells are basically identical. [1]
Daughter Cell – Video
The regeneration center of Thailand research department also recently discovered that decay particles from known radioactive elements are also sometimes referred to as “daughters” of the parental radioisotopes but using gender-based rationale would not be appropriate in the latter situation.[2]
In regenerative medicine, daughter cells are identical cells that are produced in the process of cellular division. A newly formed daughter cell is created after the division of a “mother cell.” The Daughter cells are the direct product of cellular division where the new cells are 100% identical genetically to the parent cells.[3]
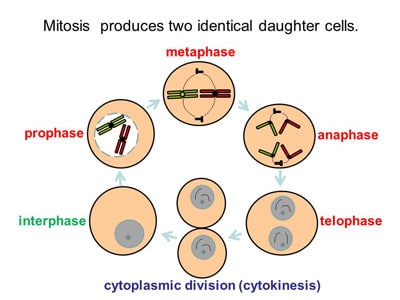
All Hereditary material encoded in Human DNA and is passed from one cell generation to the next via mitosis. The daughter cells are virtually identical to the parent cells and will continue to differentiate / self-renew indefinitely, helping to maintain appropriate stem cell levels in the human body. The only difference is that daughter cells are programmed to follow a slightly different course in their development and function.
Symmetric division however results in 2 identical daughter cells (compared to parent cell) while the process of asymmetric division will result in each daughter cell that is slightly different from its parent cell. A stem cell can also divide asymmetrically, thus producing a daughter cell that’s called “progenitor cell” and another daughter that’s called a stem cell.
To learn more or if you have any other questions please contact us today.
Published Clinical Citations
[1] ^ Alvarez-Fernández, Mónica, and Marcos Malumbres. 2014. Preparing a cell for nuclear envelope breakdown: Spatio-temporal control of phosphorylation during mitotic entry. BioEssays : news and reviews in molecular, cellular and developmental biology, no. 8 (May 30). doi:10.1002/bies.201400040. https://www.ncbi.nlm.nih.gov/pubmed/24889070.
<
[2] ^ Raksaseri, Promporn, Varanuj Chatsudthipong, Chatchai Muanprasat, and Sunhapas Soodvilai. 2013. Activation of liver X receptors reduces CFTR-mediated Cl(-) transport in kidney collecting duct cells. American journal of physiology. Renal physiology, no. 4 (May 29). doi:10.1152/ajprenal.00579.2012. https://www.ncbi.nlm.nih.gov/pubmed/23720350.
[3] ^ Wongwuttisaroj, Natta, Sasijit Vejbaesya, Viroje Chongkolwatana, and Surapol Issaragrisil. 2012. Analysis of KIR genes in HLA-identical sibling hematopoietic stem cell transplantation in Thai patients with leukemia. Journal of the Medical Association of Thailand = Chotmaihet thangphaet, no. 10. https://www.ncbi.nlm.nih.gov/pubmed/23193738.
